

 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data
| Target/Host (Institution) | Ligand | Target/Host Links | Ligand Links | Trg + Lig Links | Ki nM | ΔG° kJ/mole | IC50 nM | Kd nM | EC50/IC50 nM | koff s-1 | kon M-1s-1 | pH | Temp °C |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Vitamin D3 receptor A (Danio rerio) | BDBM50241974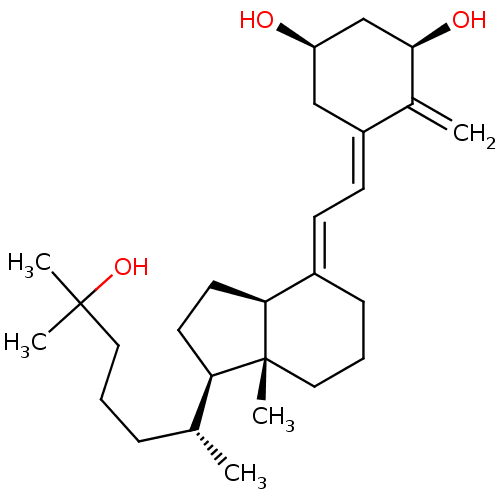 (CHEMBL4099875) | PDB UniProtKB/SwissProt GoogleScholar AffyNet | Purchase CHEMBL MCE PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | n/a | n/a | 0.140 | n/a | n/a | n/a | n/a |
IGBMC (Institute of Genetics and of Molecular and Cellular Biology) Curated by ChEMBL | Assay Description Agonist activity at zebrafish VDR LBD (156 to 453 residues) expressed in HEK293 EBNA cells harboring human CYP24-Tk-Luc plasmid assessed as induction... | Eur J Med Chem 134: 86-96 (2017) Article DOI: 10.1016/j.ejmech.2017.03.081 BindingDB Entry DOI: 10.7270/Q2N018QM | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Vitamin D3 receptor A (Danio rerio) | BDBM50241975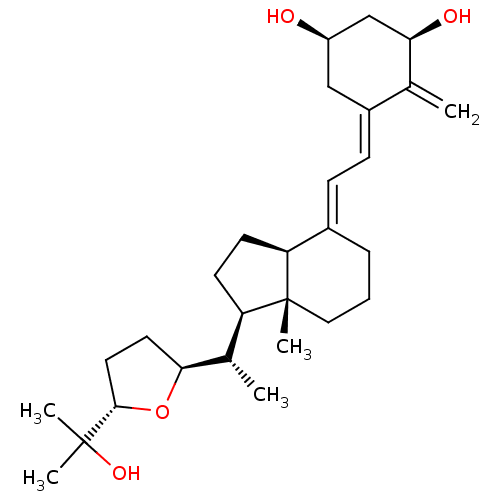 (CHEMBL4060864) | PDB UniProtKB/SwissProt GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | n/a | n/a | >100 | n/a | n/a | n/a | n/a |
IGBMC (Institute of Genetics and of Molecular and Cellular Biology) Curated by ChEMBL | Assay Description Agonist activity at zebrafish VDR LBD (156 to 453 residues) expressed in HEK293 EBNA cells harboring human CYP24-Tk-Luc plasmid assessed as induction... | Eur J Med Chem 134: 86-96 (2017) Article DOI: 10.1016/j.ejmech.2017.03.081 BindingDB Entry DOI: 10.7270/Q2N018QM | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Vitamin D3 receptor A (Danio rerio) | BDBM50241976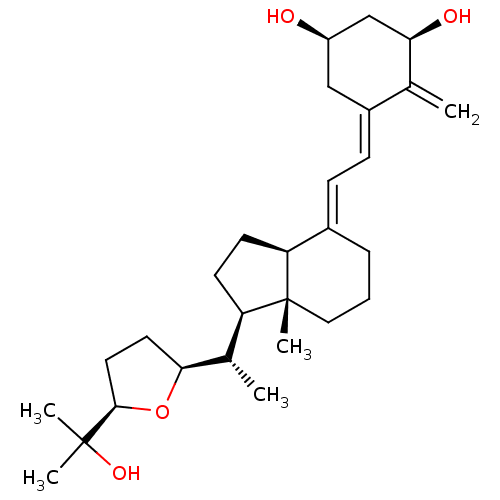 (CHEMBL4088053) | PDB UniProtKB/SwissProt GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | n/a | n/a | >100 | n/a | n/a | n/a | n/a |
IGBMC (Institute of Genetics and of Molecular and Cellular Biology) Curated by ChEMBL | Assay Description Agonist activity at zebrafish VDR LBD (156 to 453 residues) expressed in HEK293 EBNA cells harboring human CYP24-Tk-Luc plasmid assessed as induction... | Eur J Med Chem 134: 86-96 (2017) Article DOI: 10.1016/j.ejmech.2017.03.081 BindingDB Entry DOI: 10.7270/Q2N018QM | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Vitamin D3 receptor A (Danio rerio) | BDBM50241977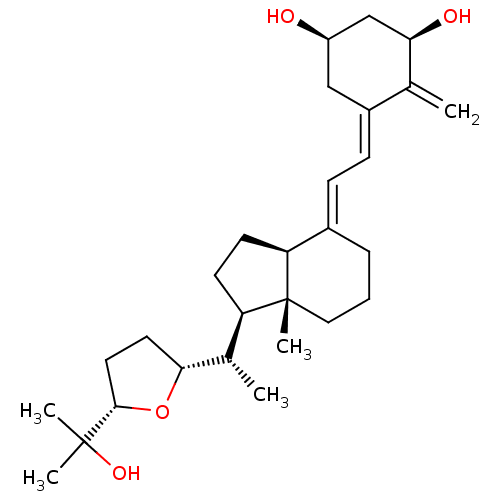 (CHEMBL4066758) | PDB UniProtKB/SwissProt GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | n/a | n/a | >100 | n/a | n/a | n/a | n/a |
IGBMC (Institute of Genetics and of Molecular and Cellular Biology) Curated by ChEMBL | Assay Description Agonist activity at zebrafish VDR LBD (156 to 453 residues) expressed in HEK293 EBNA cells harboring human CYP24-Tk-Luc plasmid assessed as induction... | Eur J Med Chem 134: 86-96 (2017) Article DOI: 10.1016/j.ejmech.2017.03.081 BindingDB Entry DOI: 10.7270/Q2N018QM | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Vitamin D3 receptor A (Danio rerio) | BDBM50496699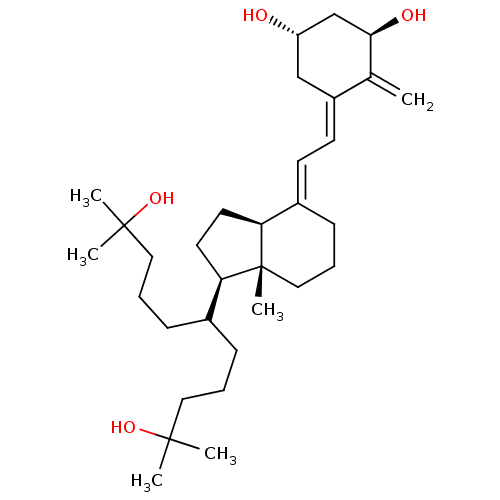 (CHEMBL3220716) | PDB UniProtKB/SwissProt GoogleScholar AffyNet | PC cid PC sid UniChem | Article PubMed | n/a | n/a | n/a | n/a | 1.20 | n/a | n/a | n/a | n/a |
Centre National de la Recherche Scientifique Curated by ChEMBL | Assay Description Agonist activity at zebrafish gal4-VDR LBD expressed in human MCF7 cells by luciferase reporter gene based transactivation assay | Medchemcomm 2: 424-429 (2011) Article DOI: 10.1039/c1md00059d BindingDB Entry DOI: 10.7270/Q29W0JFT | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Vitamin D3 receptor A (Danio rerio) | BDBM50200182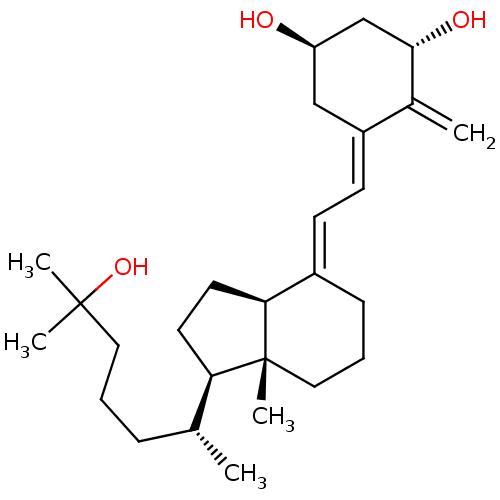 ((1S,3R,5Z,7E)-9,10-secocholesta-5,7,10(19)-triene-...) | PDB UniProtKB/SwissProt GoogleScholar AffyNet | Purchase CHEMBL DrugBank MCE KEGG MMDB PC cid PC sid PDB UniChem Patents Similars | PDB Article PubMed | n/a | n/a | n/a | n/a | 5.5 | n/a | n/a | n/a | n/a |
Centre National de la Recherche Scientifique Curated by ChEMBL | Assay Description Agonist activity at zebrafish gal4-VDR LBD expressed in human MCF7 cells by luciferase reporter gene based transactivation assay | Medchemcomm 2: 424-429 (2011) Article DOI: 10.1039/c1md00059d BindingDB Entry DOI: 10.7270/Q29W0JFT | |||||||||||
| More data for this Ligand-Target Pair |  3D Structure (crystal) | ||||||||||||
| Vitamin D3 receptor A (Danio rerio) | BDBM50409927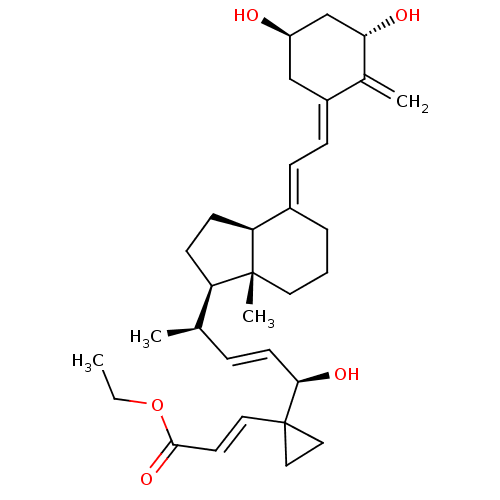 (CHEMBL2112315) | PDB UniProtKB/SwissProt GoogleScholar AffyNet | Purchase CHEMBL MCE PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | n/a | >3.00E+4 | n/a | n/a | n/a | n/a | n/a |
Institut de G£n£tique et de Biologie Mol£culaire et Cellulaire (IGBMC) Curated by ChEMBL | Assay Description Binding affinity to zebrafish VDR LBD assessed as Kd for fluorescein-labeled SRC1 NR2 peptide binding by micro-scale thermophoresis method | J Med Chem 63: 9457-9463 (2020) Article DOI: 10.1021/acs.jmedchem.0c00656 BindingDB Entry DOI: 10.7270/Q28919DP | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Vitamin D3 receptor A (Danio rerio) | BDBM50496700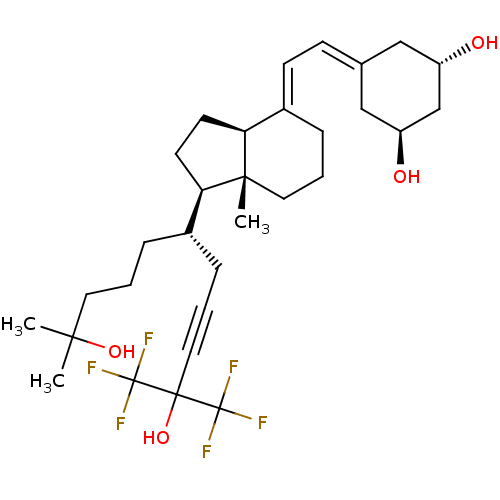 (CHEMBL3220717) | PDB UniProtKB/SwissProt GoogleScholar AffyNet | PC cid PC sid UniChem | Article PubMed | n/a | n/a | n/a | 1.80E+3 | n/a | n/a | n/a | n/a | n/a |
Centre National de la Recherche Scientifique Curated by ChEMBL | Assay Description Agonist activity at zebrafish VDR LBD assessed as binding affinity to TAMRA-labeled SRC-1 peptide by fluorescence anisotropy assay | Medchemcomm 2: 424-429 (2011) Article DOI: 10.1039/c1md00059d BindingDB Entry DOI: 10.7270/Q29W0JFT | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Vitamin D3 receptor A (Danio rerio) | BDBM50496701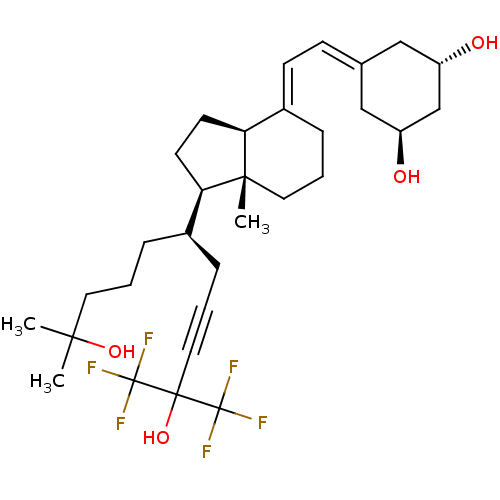 (CHEMBL3220718) | PDB UniProtKB/SwissProt GoogleScholar AffyNet | PC cid PC sid UniChem | Article PubMed | n/a | n/a | n/a | 1.80E+3 | n/a | n/a | n/a | n/a | n/a |
Centre National de la Recherche Scientifique Curated by ChEMBL | Assay Description Agonist activity at zebrafish VDR LBD assessed as binding affinity to TAMRA-labeled SRC-1 peptide by fluorescence anisotropy assay | Medchemcomm 2: 424-429 (2011) Article DOI: 10.1039/c1md00059d BindingDB Entry DOI: 10.7270/Q29W0JFT | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Vitamin D3 receptor A (Danio rerio) | BDBM50496701 (CHEMBL3220718) | PDB UniProtKB/SwissProt GoogleScholar AffyNet | PC cid PC sid UniChem | Article PubMed | n/a | n/a | n/a | n/a | 0.25 | n/a | n/a | n/a | n/a |
Centre National de la Recherche Scientifique Curated by ChEMBL | Assay Description Agonist activity at zebrafish gal4-VDR LBD expressed in human MCF7 cells by luciferase reporter gene based transactivation assay | Medchemcomm 2: 424-429 (2011) Article DOI: 10.1039/c1md00059d BindingDB Entry DOI: 10.7270/Q29W0JFT | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Vitamin D3 receptor A (Danio rerio) | BDBM50496699 (CHEMBL3220716) | PDB UniProtKB/SwissProt GoogleScholar AffyNet | PC cid PC sid UniChem | Article PubMed | n/a | n/a | n/a | 3.30E+3 | n/a | n/a | n/a | n/a | n/a |
Centre National de la Recherche Scientifique Curated by ChEMBL | Assay Description Agonist activity at zebrafish VDR LBD assessed as binding affinity to TAMRA-labeled SRC-1 peptide by fluorescence anisotropy assay | Medchemcomm 2: 424-429 (2011) Article DOI: 10.1039/c1md00059d BindingDB Entry DOI: 10.7270/Q29W0JFT | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Vitamin D3 receptor A (Danio rerio) | BDBM50200182 ((1S,3R,5Z,7E)-9,10-secocholesta-5,7,10(19)-triene-...) | PDB UniProtKB/SwissProt GoogleScholar AffyNet | Purchase CHEMBL DrugBank MCE KEGG MMDB PC cid PC sid PDB UniChem Patents Similars | PDB Article PubMed | n/a | n/a | n/a | 1.20E+3 | n/a | n/a | n/a | n/a | n/a |
Institut de G£n£tique et de Biologie Mol£culaire et Cellulaire (IGBMC) Curated by ChEMBL | Assay Description Binding affinity to zebrafish VDR LBD assessed as Kd for fluorescein-labeled SRC1 NR2 peptide binding by micro-scale thermophoresis method | J Med Chem 63: 9457-9463 (2020) Article DOI: 10.1021/acs.jmedchem.0c00656 BindingDB Entry DOI: 10.7270/Q28919DP | |||||||||||
| More data for this Ligand-Target Pair |  3D Structure (crystal) | ||||||||||||
| Vitamin D3 receptor A (Danio rerio) | BDBM50496700 (CHEMBL3220717) | PDB UniProtKB/SwissProt GoogleScholar AffyNet | PC cid PC sid UniChem | Article PubMed | n/a | n/a | n/a | n/a | 0.150 | n/a | n/a | n/a | n/a |
Centre National de la Recherche Scientifique Curated by ChEMBL | Assay Description Agonist activity at zebrafish gal4-VDR LBD expressed in human MCF7 cells by luciferase reporter gene based transactivation assay | Medchemcomm 2: 424-429 (2011) Article DOI: 10.1039/c1md00059d BindingDB Entry DOI: 10.7270/Q29W0JFT | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||